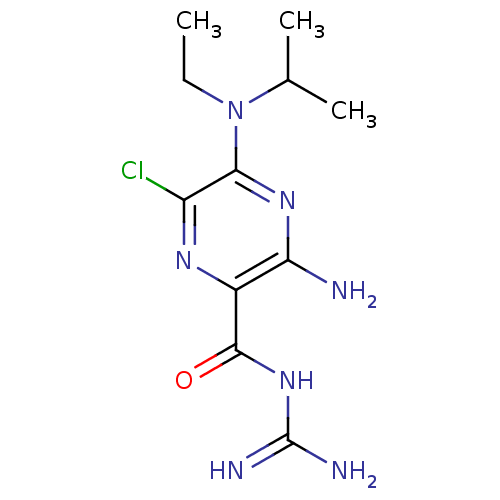
 CHEMBL1909809 CHEMBL517986 cid_1795 BDBM50270005 5'-(N-ethyl-N-isopropyl)amiloride
CHEMBL1909809 CHEMBL517986 cid_1795 BDBM50270005 5'-(N-ethyl-N-isopropyl)amiloride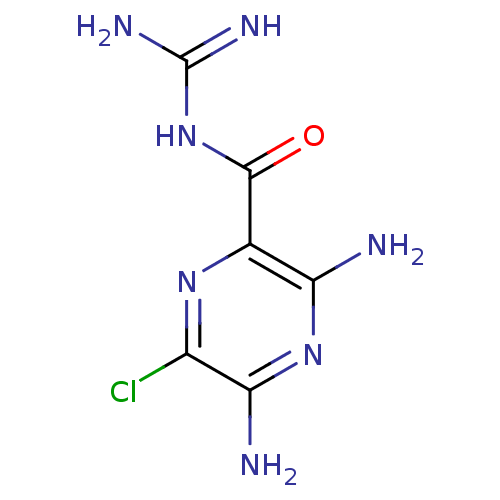 Amipramidin Amiloride CHEMBL945 3,5-diamino-N-carbamimidoyl-6-chloropyrazine-2-carboxamide 3,5-diamino-6-chloro-N-(diaminomethylene)pyrazinamide;hydrochloride BDBM16173
Amipramidin Amiloride CHEMBL945 3,5-diamino-N-carbamimidoyl-6-chloropyrazine-2-carboxamide 3,5-diamino-6-chloro-N-(diaminomethylene)pyrazinamide;hydrochloride BDBM16173
- Buckley, BJ; Aboelela, A; Majed, H; Bujaroski, RS; White, KL; Powell, AK; Wang, W; Katneni, K; Saunders, J; Shackleford, DM; Charman, SA; Cook, GM; Kelso, MJ; Ranson, M Systematic evaluation of structure-property relationships and pharmacokinetics in 6-(hetero)aryl-substituted matched pair analogs of amiloride and 5-(N,N-hexamethylene)amiloride. Bioorg Med Chem 37: (2021)
- Kuduk, SD; Di Marco, CN; Chang, RK; Dipardo, RM; Cook, SP; Cato, MJ; Jovanovska, A; Urban, MO; Leitl, M; Spencer, RH; Kane, SA; Bilodeau, MT; Hartman, GD; Bock, MG Amiloride derived inhibitors of acid-sensing ion channel-3 (ASIC3). Bioorg Med Chem Lett 19: 2514-8 (2009)
- Massey, AP; Harley, WR; Pasupuleti, N; Gorin, FA; Nantz, MH 2-Amidino analogs of glycine-amiloride conjugates: inhibitors of urokinase-type plasminogen activator. Bioorg Med Chem Lett 22: 2635-9 (2012)
- Matthews, H; Ranson, M; Tyndall, JD; Kelso, MJ Synthesis and preliminary evaluation of amiloride analogs as inhibitors of the urokinase-type plasminogen activator (uPA). Bioorg Med Chem Lett 21: 6760-6 (2011)
- Buckley, BJ; Majed, H; Aboelela, A; Minaei, E; Jiang, L; Fildes, K; Cheung, CY; Johnson, D; Bachovchin, D; Cook, GM; Huang, M; Ranson, M; Kelso, MJ 6-Substituted amiloride derivatives as inhibitors of the urokinase-type plasminogen activator for use in metastatic disease. Bioorg Med Chem Lett 29: (2019)
- Massink, A; Louvel, J; Adlere, I; van Veen, C; Huisman, BJ; Dijksteel, GS; Guo, D; Lenselink, EB; Buckley, BJ; Matthews, H; Ranson, M; Kelso, M; IJzerman, AP 5'-Substituted Amiloride Derivatives as Allosteric Modulators Binding in the Sodium Ion Pocket of the Adenosine A2A Receptor. J Med Chem 59: 4769-77 (2016)
- Buckley, BJ; Aboelela, A; Minaei, E; Jiang, LX; Xu, Z; Ali, U; Fildes, K; Cheung, CY; Cook, SM; Johnson, DC; Bachovchin, DA; Cook, GM; Apte, M; Huang, M; Ranson, M; Kelso, MJ 6-Substituted Hexamethylene Amiloride (HMA) Derivatives as Potent and Selective Inhibitors of the Human Urokinase Plasminogen Activator for Use in Cancer. J Med Chem 61: 8299-8320 (2018)
- S El Salamouni, N; Buckley, BJ; Jiang, L; Huang, M; Ranson, M; Kelso, MJ; Yu, H Disruption of Water Networks is the Cause of Human/Mouse Species Selectivity in Urokinase Plasminogen Activator (uPA) Inhibitors Derived from Hexamethylene Amiloride (HMA). J Med Chem 65: 1933-1945 (2022)
- Pedersen, SF; King, SA; Nygaard, EB; Rigor, RR; Cala, PM NHE1 inhibition by amiloride- and benzoylguanidine-type compounds. Inhibitor binding loci deduced from chimeras of NHE1 homologues with endogenous differences in inhibitor sensitivity. J Biol Chem 282: 19716-27 (2007)
- Garritsen, A; Ijzerman, AP; Tulp, MT; Cragoe, EJ; Soudijn, W Receptor binding profiles of amiloride analogues provide no evidence for a link between receptors and the Na+/H+ exchanger, but indicate a common structure on receptor proteins. J Recept Res 11: 891-907 (1991)
- ChEMBL_164985 (CHEMBL774080) Inhibition of 5-N-(ethyl isopropyl)-amiloride[EIPA]-sensitive Na+ uptake into acidified rabbit erythrocytes.
- ChEMBL_2152472 (CHEMBL5037019) Potentiation of wild type CFTR (unknown origin) expressed in HBE cells assessed as change in short circuit current in presence of forskolin and amiloride
- ChEMBL_2152470 (CHEMBL5037017) Potentiation of CFTR F508 deletion mutant (unknown origin) expressed in HBE cells assessed as change in short circuit current in presence of forskolin and amiloride
- ChEMBL_2222625 (CHEMBL5135959) Modulation of CFTR F508 deletion mutant expressed in HBE cells assessed as change in short circuit current incubated for 18 to 24 hrs in presence of forskolin, amiloride and human serum by voltage-clamp based chamber assay
- ChEMBL_2235954 (CHEMBL5149726) Inhibition of human NHE3 expressed in opossum kidney cells assessed as reduction in Na-HEPES buffer-mediated pH recovery in presence of NEH1 inhibitor ethyl isopropyl amiloride by BCECF-AM dye based fluorescence assay
- ChEMBL_2235955 (CHEMBL5149727) Inhibition of rat NHE3 expressed in opossum kidney cells assessed as reduction in Na-HEPES buffer-mediated pH recovery in presence of NEH1 inhibitor ethyl isopropyl amiloride by BCECF-AM dye based fluorescence assay
- In Vitro Measure of Sodium Channel Blocking Activity and Reversibility One assay used to assess mechanism of action and/or potency of the compounds of the present invention involves the determination of lumenal drug inhibition of airway epithelial sodium currents measured under short circuit current (ISC) using airway epithelial monolayers mounted in Using chambers. This assay is described in detail in Hirsh, A. J., Zhang, J., Zamurs, A., et al. Pharmacological properties of N-(3,5-diamino-6-chloropyrazine-2-carbonyl)-N′-4-[4-(2,3-dihydroxypropoxy)phenyl]butyl-guanidine methanesulfonate (552-02), a novel epithelial sodium channel blocker with potential clinical efficacy for CF lung disease. J. Pharmacol. Exp. Ther. 2008; 325(1): 77-88. Cells obtained from freshly excised human, dog, sheep or rodent airways are seeded onto porous 0.4 micron Snapwell Inserts (CoStar), cultured at air-liquid interface (ALI) conditions in hormonally defined media, and assayed for sodium transport activity (ISC) while bathed in Krebs Bicarbonate Ringer (KBR) in Using chambers. All test drug additions are to the lumenal bath with half-log dose addition protocols (from 1�10−11 M to 3�10−5 M), and the cumulative change in ISC (inhibition) recorded. All drugs are prepared in dimethyl sulfoxide as stock solutions at a concentration of 1�10−2 M and stored at −20� C. Eight preparations are typically run in parallel; two preparations per run incorporate amiloride and/or benzamil as positive controls. After the maximal concentration (5�10−5 M) is administered, the lumenal bath is exchanged three times with fresh drug-free KBR solution, and the resultant ISC measured after each wash for approximately 5 minutes in duration. Reversibility is defined as the percent return to the baseline value for sodium current after the third wash. All data from the voltage clamps are collected via a computer interface and analyzed off-line.Dose-effect relationships for all compounds are considered and analyzed by the Prism 3.0 program. IC50 values, maximal effective concentrations, and reversibility are calculated and compared to amiloride and benzamil as positive controls.
- Chamber Electrophysiology Assay of CFTR Potentiation in CF Bronchial Epithelial Cells Primary cystic fibrosis human bronchial epithelial (CF hBE) cells were expanded and cultured according to published methods (Neuberger et al., Ch. 4 of Cystic Fibrosis, Methods in Molecular Biology vol. 741, pp. 39-54 (2011)). Well-differentiated cells (>30 days at air/liquid interface) on Snapwell filters (Corning Costar, cat. no. 3801) were mounted in Ussing chambers (Physiologic Instruments, Inc., San Diego, Calif.). F508del/F508del cultures were assayed at 27� C. and G551D/F508del cells were assayed at 35� C. HEPES buffered physiological saline (composition (in mM): 137 NaCl, 4 KCl, 1 MgCl2, 1.8 CaCl2, 10 HEPES Na) was used in both apical and basolateral chambers. Chambers were bubbled with air to promote mixing and the voltage was clamped to zero. Amiloride (30 uM), forskolin (10 uM), test compound (4 increasing concentrations), and CFTRinh-172 (20 uM) were added sequentially with 20-25 minutes between additions. Short-circuit currents were acquired and analyzed using LabScribe2. Test compound responses were scaled relative to responses for DMSO (0%) and the maximal response of a positive control potentiator (100%).
- Chamber Assay of CFTR-mediated short-circuit currents ssing chamber experiments were performed using human bronchial epithelial (HBE) cells derived from CF subjects heterozygous for F508del and a minimal function CFTR mutation (F508del/MF-HBE) and cultured as previously described (Neuberger T, Burton B, Clark H, Van Goor F Methods Mol Biol 2011:741:39-54). After four days the apical media was removed, and the cells were grown at an air liquid interface for >14 days prior to use. This resulted in a monolayer of fully differentiated columnar cells that were ciliated, features that are characteristic of human bronchial airway epithelia. To isolate the CFTR-mediated short-circuit (ISC) current, F508del/MF-HBE grown on Costar Snapwell cell culture inserts were mounted in an Ussing chamber and the transepithelial ISC was measured under voltage-clamp recording conditions (Vhold= 0 mV) at 37 oC. The basolateral solution contained (in mM) 145 NaCl, 0.83 K2HPO4, 3.3 KH2PO4, 1.2 MgCl2, 1.2 CaCl2, 10 Glucose, 10 HEPES (pH adjusted to 7.4 with NaOH) and the apical solution contained (in mM) 145 NaGluconate, 1.2 MgCl2, 1.2 CaCl2, 10 glucose, 10 HEPES (pH adjusted to 7.4 with NaOH) and 30 �M amiloride to block the epithelial sodium channel. Forskolin (20 �M) was added to the apical surface to activate CFTR, followed by apical addition of a CFTR inhibitor cocktail consisting of BPO, GlyH-101 and CFTR inhibitor 172 (each at 20 �M final assay concentration) to specifically isolate CFTR currents. The CFTR-mediated ISC (�A/cm2) for each condition was determined from the peak forskolin response to the steady-state current following inhibition.
- Cell-Based Assay of NHE-3 Activity Rat NHE-3-mediated Na+-dependent H+ antiport was measured using a modification of the pH sensitive dye method originally reported by Tsien (Proc. Natl. Acad. Sci. USA. (1984) 81(23): 7436-7440). Opossum kidney (OK) cells were obtained from the ATCC and propagated per their instructions. The rat NHE-3 gene was introduced into OK cells via electroporation, seeded into 96 well plates and grown overnight. Medium was aspirated from the wells, cells were washed twice with NaCl-HEPES buffer (100 mM NaCl, 50 mM HEPES, 10 mM glucose, 5 mM KCl, 2 mM CaCl2, 1 mM MgCl2, pH 7.4), then incubated for 30 min at room temperature with NH4Cl-HEPES buffer (20 mM NH4Cl, 80 mM NaCl, 50 mM HEPES, 5 mM KCl, 2 mM CaCl2, 1 mM MgCl2, pH 7.4) containing 5 uM BCECF-AM (Invitrogen). Cells were washed twice with Ammonium free, Na+-free HEPES (100 mM choline, 50 mM HEPES, 10 mM glucose, 5 mM KCl, 2 mM CaCl2, 1 mM MgCl2, pH 7.4) and incubated in the same buffer for 10 minutes at room temperature to lower intracellular pH. NHE-3-mediated recovery of neutral intracellular pH was initiated by addition of Na-HEPES buffer containing 5 uM ethyl isopropyl amiloride (EIPA, a selective antagonist of NHE-1 activity that does not inhibit NHE-3) and 0-30 uM test compound, and monitoring the pH sensitive changes in BCECF fluorescence (ex 505 nm, λem 538 nm) normalized to the pH insensitive BCECF fluorescence (λex 439 nm, λem 538 nm). Initial rates were were plotted as the average 3-6 replicates, and pIC50 values were estimated using GraphPad Prism.
- Cell-Based Assay of NHE-3 Activity (Pre-Incubation Inhibition) Rat and human NHE-3-mediated Na+-dependent H+ antiport was measured using a modification of the pH sensitive dye method originally reported by Paradiso (Proc. Natl. Acad. Sci. USA. (1984) 81(23): 7436-7440). PS120 fibroblasts stably expressing human NHE3 and NHERF2 were obtained from Mark Donowitz (Baltimore, Md.). Opossum kidney (OK) cells were obtained from the ATCC and propagated per their instructions. The rat NHE-3 gene (GenBank M85300) was introduced into OK cells via electroporation, and cells were seeded into 96 well plates and grown overnight. Medium was aspirated from the wells then incubated for 30 min at 37� C. with NH4Cl-HEPES buffer (20 mM NH4Cl, 80 mM NaCl, 50 mM HEPES, 5 mM KCl, 2 mM CaCl2, 1 mM MgCl2, pH 7.4) containing 5 μM BCECF-AM. Cells were washed once with Ammonium free, Na+-free HEPES (100 mM choline, 50 mM HEPES, 10 mM glucose, 5 mM KCl, 2 mM CaCl2, 1 mM MgCl2, pH 7.4) and incubated in the same buffer for 10 minutes at room temperature to lower intracellular pH with 0-30 μM test compound. After incubation, NHE-3-mediated recovery of neutral intracellular pH was initiated by addition of Na-HEPES buffer containing 0.4 μM ethyl isopropyl amiloride (EIPA, a selective antagonist of NHE-1 activity that does not inhibit NHE-3). Changes in intracellular pH were monitored using a FLIPR Tetra (Molecular Devices, Sunnyvale, Calif.) by excitation at λex 439 to 505 nm, and measuring BCECF fluorescence at λem 538 nm. The initial rate of the fluorescence ratio change was used as a measure of NHE-mediated Na+/H+ activity, and reported as the change in fluorescence ratio per minute. Initial rates were plotted as the average of 2 or more replicates, and pIC50 values were estimated using GraphPad Prism.
- Cell-Based Assay of NHE3 Activity under Persistent Conditions The ability of compounds to inhibit Rat NHE3-mediated Na+-dependent H+ antiport after application and washout was measured using a modification of the pH sensitive dye method described above. Opossum kidney (OK) cells were obtained from the ATCC and propagated per their instructions. The rat NHE3 gene was introduced into OK cells via electroporation, and cells were seeded into 96 well plates and grown overnight. Medium was aspirated from the wells, cells were washed twice with NaCl-HEPES buffer (100 mM NaCl, 50 mM HEPES, 10 mM glucose, 5 mM KCl, 2 mM CaCl2, 1 mM MgCl2, pH 7.4), then overlayed with NaCl-HEPES buffer containing 0-30 μM test compound.After a 60 min incubation, the test drug containing buffer was aspirated from the cells, cells were washed twice with NaCl-HEPES buffer without drug, then incubated for 30 min at room temperature with NH4Cl-HEPES buffer (20 mM NH4Cl, 80 mM NaCl, 50 mM HEPES, 5 mM KCl, 2 mM CaCl2, 1 mM MgCl2, pH 7.4) containing 5 uM BCECF-AM. Cells were washed twice with Ammonium free, Natfree HEPES (100 mM choline, 50 mM HEPES, 10 mM glucose, 5 mM KCl, 2 mM CaCl2, 1 mM MgCl2, pH 7.4) and incubated in the same buffer for 10 minutes at room temperature to lower intracellular pH. NHE3-mediated recovery of neutral intracellular pH was initiated (40 min after compound washout) by addition of Na-HEPES buffer containing 0.4 uM ethyl isopropyl amiloride (EIPA, a selective antagonist of NHE-1 activity that does not inhibit NHE3), and monitoring the pH sensitive changes in BCECF fluorescence (λex 505 nm, λem, 538 nm) normalized to the pH insensitive BCECF fluorescence (λex 439 nm, λem, 538 nm). Initial rates were plotted as the average 2 or more replicates, and pIC50 values w
- Cell-Based Assay of NHE-3 Activity (Persistent Inhibition) The ability of compounds to inhibit human and rat NHE-3-mediated Na+ dependent H+ antiport after application and washout was measured using a modification of the pH sensitive dye method described above. PS120 fibroblasts stably expressing human NHE3 and NHERF2 were obtained from Mark Donowitz (Baltimore, Md.). Opossum kidney (OK) cells were obtained from the ATCC and propagated per their instructions. The rat NHE-3 gene was introduced into OK cells via electroporation, and cells were seeded into 96 well plates and grown overnight. Medium was aspirated from the wells, cells were washed once with NaCl-HEPES buffer (100 mM NaCl, 50 mM HEPES, 10 mM glucose, 5 mM KCl, 2 mM CaCl2, 1 mM MgCl2, pH 7.4), then overlayed with NaCl-HEPES buffer containing 0-30 μM test compound. After a 60 min incubation at room temperature, the test drug containing buffer was aspirated from the cells. Following aspiration, cells were washed once with NaCl-HEPES buffer without drug, then incubated for 30 min at 37� C. with NH4Cl-HEPES buffer (20 mM NH4Cl, 80 mM NaCl, 50 mM HEPES, 5 mM KCl, 2 mM CaCl2, 1 mM MgCl2, pH 7.4) containing 5 μM BCECF-AM. Cells were washed once with Ammonium free, Na+-free HEPES (100 mM choline, 50 mM HEPES, 10 mM glucose, 5 mM KCl, 2 mM CaCl2, 1 mM MgCl2, pH 7.4) and incubated in the same buffer for 10 minutes at room temperature to lower intracellular pH. NHE-3-mediated recovery of neutral intracellular pH was initiated (10 min after compound washout) by addition of Na-HEPES buffer. For the rat NHE3 assay, the Na-HEPES buffer contained 0.4 ethyl isopropyl amiloride (EIPA, a selective antagonist of NHE-1 activity that does not inhibit NHE-3). Changes in intracellular pH were monitored using a FLIPR Tetra (Molecular Devices, Sunnyvale, Calif.) by excitation at λex 439 to 505 nm, and measuring BCECF fluorescence at λem 538 nm. The initial rate of the fluorescence ratio change was used as a measure of NHE-mediated Na+/H+ activity, and reported as the change in fluorescence ratio per minute. Initial rates were plotted as the average of 2 or more replicates, and pIC50 values were estimated using GraphPad Prism.
- Cell-based activity under Prompt Conditions Cell-based activity under Prompt Conditions. Rat or human NHE3-mediated Na+-dependent H+ antiport was measured using a modification of the pH sensitive dye method originally reported by Paradiso (PNAS USA. 81:7436-7440, 1984). Opossum kidney (OK) cells were obtained from the ATCC and propagated per their instructions. The rat NHE3 gene (GenBank M85300) or the human NHE3 gene (GenBank NM_004174.1) was introduced into OK cells via electroporation, and cells were seeded into 96 well plates and grown overnight. Medium was aspirated from the wells, cells were washed twice with NaCl-HEPES buffer (100 mM NaCl, 50 mM HEPES, 10 mM glucose, 5 mM KCl, 2 mM CaCl2, 1 mM MgCl2, pH 7.4), then incubated for 30 min at room temperature with NH4Cl-HEPES buffer (20 mM NH4Cl, 80 mM NaCl, 50 mM HEPES, 5 mM KCl, 2 mM CaCl2, 1 mM MgCl2, pH 7.4) containing 5 μM bis(acetoxymethyl) 3,3′-(3′,6′-bis(acetoxymethoxy)-5-((acetoxymethoxy)carbonyl)-3-oxo-3H-spiro[isobenzofuran-1,9′-xanthene]-2′,7′-diyl)dipropanoate (BCECF-AM).Cells were washed twice with Ammonium free, Na+-free HEPES (100 mM choline, 50 mM HEPES, 10 mM glucose, 5 mM KCl, 2 mM CaCl2, 1 mM MgCl2, pH 7.4) and incubated in the same buffer for 10 minutes at room temperature to lower intracellular pH. NHE3-mediated recovery of neutral intracellular pH was initiated by addition of Na-HEPES buffer containing 0.4 μM ethyl isopropyl amiloride (EIPA, a selective antagonist of NHE-1 activity that does not inhibit NHE3) and 0-30 μM test compound, or a pharmaceutically acceptable salt thereof, and monitoring the pH sensitive changes in BCECF fluorescence (λex 505 nm, λem, 538 nm) normalized to the pH insensitive BCECF fluorescence (λex 439 nm, λem, 538 nm). Initial rates were plotted as the average 2 or more replicates, and pIC50 values were estimated using GraphPad Prism.

